Phone: 1-888-323-5131 (toll free US only)
Phone: 1-215-526-2180 (worldwide)
Fax: 215-526-2191
Email: info@verigenics.com
Hose Track® RF Identification and Hose Lifecycle Analysis System
![]()

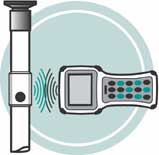
 Hose Track® offers a new degree of safety and reduced risk for the pharmaceutical processing industry, as well as other high purity applications in the food and beverage, cosmetic, chemical, and biomedical sectors. Using RFID (radio frequency identification) technology, all process equipment involved with a particular batch of product is monitored from start to finish. Briefly stated, Hose Track tracks who did what to each batch and when. It's a logical approach to tracking all critical process components.
Hose Track® offers a new degree of safety and reduced risk for the pharmaceutical processing industry, as well as other high purity applications in the food and beverage, cosmetic, chemical, and biomedical sectors. Using RFID (radio frequency identification) technology, all process equipment involved with a particular batch of product is monitored from start to finish. Briefly stated, Hose Track tracks who did what to each batch and when. It's a logical approach to tracking all critical process components.
HOSE TRACK HIGHLIGHTS
- Identifies individual process equipment devices such as hoses, pumps, bio-bags, diaphragm valves, filters, and UV lamps using RFID tags
- Tracks key wear-related events such as cleaning cycles/dates and batches of material processed or other user-defined events
- Ensures timely maintenance and replacement before parts begin to fail, risk product integrity, and waste time and labor
- Provides an audit trail to assist in validation processes; helps accelerate data collection procedures and lower costs
- Reduces errors on the production floor
- Consolidates documents by electronically linking to notes, cleaning schedules, files, certifications, photos and illustrations, installation instructions, warning notices, and other protocols
- Safe for use with CIP, SIP, and autoclave processes
- Various tags address applications involving high temperatures, gamma irradiation, and mounting to metal
- Gamma-radiation-resistant RFID tag – GammaTag® – now available
- Different tag sizes and shapes available
- Field installation methods available to immediately start tracking existing equipment
- U.S. Patents 7,195,149; 7,328,837 and 8,519,846
ADVANTAGES
Minimizes Risk
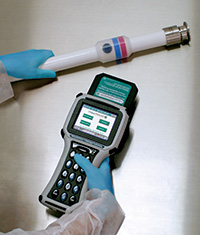
- Tags are encoded with serial numbers and other information and are externally attached to each piece of process equipment – no contact with material flow
- An administrative level access, known as Kiosk mode, prevents changes to the system – restricts usage to help enforce 21 CFR Part 11 validation
- Identifies individual process equipment parts such as hoses, pumps, bio-bags, diaphragm valves, filters, and UV lamps using RFID tags
- A fast, efficient, and precise identification system for critical process equipment
- Eliminates dependence on manual log book record keeping, its inefficiencies and inaccuracies, and the books' contribution as a contamination source
 Reduces Costs
Reduces Costs
- Accesses and records the current status of any tagged component on the spot
- Reduces errors on the production floor
- Limits failures and helps calculate equipment life expectancy using actual data
- Eliminates the inefficient calendar method of swapping out used parts – reduces wasted production life and underutilized equipment
- Field installation methods available to immediately start tracking existing process equipment
- Applicable to predictive maintenance systems (PdM)
Consolidates Documents
- Eliminates inefficient paper labels and hang tags
- Reduces dependency on paper records and their inherent problems, such as transposed numbers, incorrect dates, handwriting legibility, misread data, and misfiled documents
- Electronically links to notes, cleaning and maintenance schedules, files, certifications, photos and illustrations, installation instructions, warning notices, and other protocols
- Allows recording and storage of all critical information
- Links to a database that contains the complete biographical history of parts
- Allows information access from any networked computer running the Hose Track program
Provides an FDA Audit Trail
 Third-party validated for 21 CFR Part 11
Third-party validated for 21 CFR Part 11- Speeds audits and lowers their costs by eliminating the time and labor of locating, retrieving, and gathering paper records and traditional log books
- Tracks key wear-related events such as cleaning cycles/dates, batches of material processed, calibration, and other user-defined events
- Provides a secure, tamper-proof record to track time, date, and operator data
Easy to Use
- Unlike bar code labels, tags do not require a clear sight line for reading/writing
- Safe for use with CIP, SIP, and autoclave processes
- Integrates with existing workflows
- Custom engineered to best meet individual needs
- Select the tag attachment method best suited for each particular item – choose from a molded pouch, lamination, watchband style encasement, silicone fusible tape, or any customized design
|
||||||||||||||||||||||||||||
Contact your Verigenics representative today to see how Hose Track can solve your needs at:
Phone: 215-526-2180 • Toll Free Phone (US only): 888-323-5131
![]() Click the link to download a printable version of our Hose Track brochure. Hose_Track.pdf (1.5MB)
Click the link to download a printable version of our Hose Track brochure. Hose_Track.pdf (1.5MB)
COMPARISON
LOG BOOK vs. Hose Track
CLICK HERE to view a comparison of the pen-and-paper Log Book method vs. the Hose Track RFID system
COMPARISON
BAR CODE vs. Hose Track
CLICK HERE to view a comparison of the Bar Code method vs. the Hose Track RFID system.
Our development of the Hose Track System has given us extensive experience with hose attachment methods. For example:

...silicone injection molded to the O.D. of a hose assembly

...wrapped silicone fusible tape for use with existing hoses
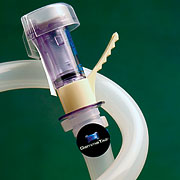
...plastic tie-wrap for use with existing hoses
NewAge Industries' AdvantaPure high purity division specialize in platinum-cured silicone tubing, biopharm grade TPE, process hoses, and molded components such as, molded manifolds and container closures for single-use systems.